Switch to List View
Image and Video Gallery
This is a searchable collection of scientific photos, illustrations, and videos. The images and videos in this gallery are licensed under Creative Commons Attribution Non-Commercial ShareAlike 3.0. This license lets you remix, tweak, and build upon this work non-commercially, as long as you credit and license your new creations under identical terms.
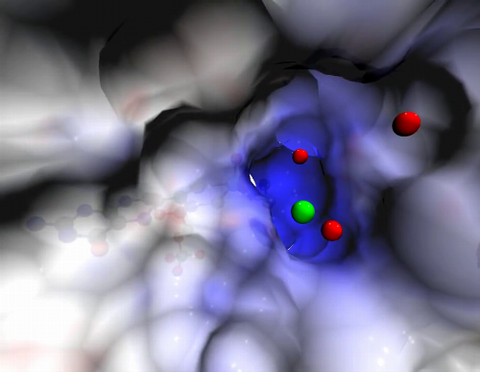
6581: Fluorescent C. elegans showing muscle and ribosomal protein
6581: Fluorescent C. elegans showing muscle and ribosomal protein
C. elegans, a tiny roundworm, with a ribosomal protein glowing red and muscle fibers glowing green. Researchers used these worms to study a molecular pathway that affects aging. The ribosomal protein is involved in protein translation and may play a role in dietary restriction-induced longevity. Image created using confocal microscopy.
View group of roundworms here 6582.
View closeup of roundworms here 6583.
View group of roundworms here 6582.
View closeup of roundworms here 6583.
Jarod Rollins, Mount Desert Island Biological Laboratory.
View Media
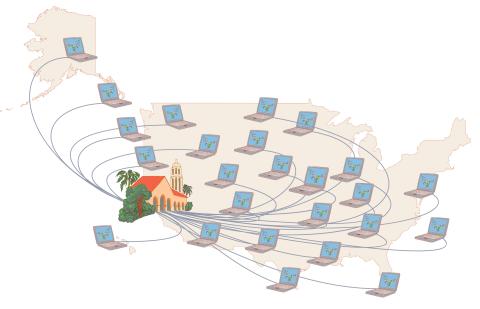
1276: Folding@Home
1276: Folding@Home
Stanford University scientist Vijay Pande decided to couple the power of computers with the help of the public. He initiated a project called Folding@Home, a so-called distributed computing project in which anyone who wants to can download a screensaver that performs protein-folding calculations when a computer is not in use. Folding@Home is modeled on a similar project called SETI@Home, which is used to search for extraterrestrial intelligence.
Judith Stoffer
View Media

2402: RNase A (2)
2402: RNase A (2)
A crystal of RNase A protein created for X-ray crystallography, which can reveal detailed, three-dimensional protein structures.
Alex McPherson, University of California, Irvine
View Media
3574: Cytonemes in developing fruit fly cells
3574: Cytonemes in developing fruit fly cells
Scientists have long known that multicellular organisms use biological molecules produced by one cell and sensed by another to transmit messages that, for instance, guide proper development of organs and tissues. But it's been a puzzle as to how molecules dumped out into the fluid-filled spaces between cells can precisely home in on their targets. Using living tissue from fruit flies, a team led by Thomas Kornberg of the University of California, San Francisco, has shown that typical cells in animals can talk to each other via long, thin cell extensions called cytonemes (Latin for "cell threads") that may span the length of 50 or 100 cells. The point of contact between a cytoneme and its target cell acts as a communications bridge between the two cells.
Sougata Roy, University of California, San Francisco
View Media
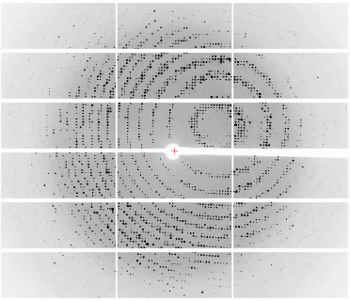
6765: X-ray diffraction pattern from a crystallized cefotaxime-CCD-1 complex
6765: X-ray diffraction pattern from a crystallized cefotaxime-CCD-1 complex
CCD-1 is an enzyme produced by the bacterium Clostridioides difficile that helps it resist antibiotics. Researchers crystallized complexes where a CCD-1 molecule and a molecule of the antibiotic cefotaxime were bound together. Then, they shot X-rays at the complexes to determine their structure—a process known as X-ray crystallography. This image shows the X-ray diffraction pattern of a complex.
Related to images 6764, 6766, and 6767.
Related to images 6764, 6766, and 6767.
Keith Hodgson, Stanford University.
View Media
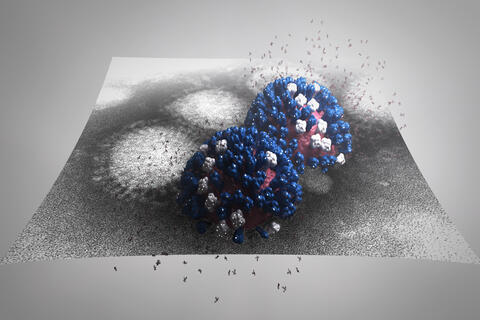
5896: Stetten Lecture 2017poster image
5896: Stetten Lecture 2017poster image
This image is featured on the poster for Dr. Rommie Amaro's 2017 Stetten Lecture. It depicts a detailed physical model of an influenza virus, incorporating information from several structural data sources. The small molecules around the virus are sialic acid molecules. The virus binds to and cleaves sialic acid as it enters and exits host cells. Researchers are building these highly detailed molecular scale models of different biomedical systems and then “bringing them to life” with physics-based methods, either molecular or Brownian dynamics simulations, to understand the structural dynamics of the systems and their complex interactions with drug or substrate molecules.
Dr. Rommie Amaro, University of California, San Diego
View Media
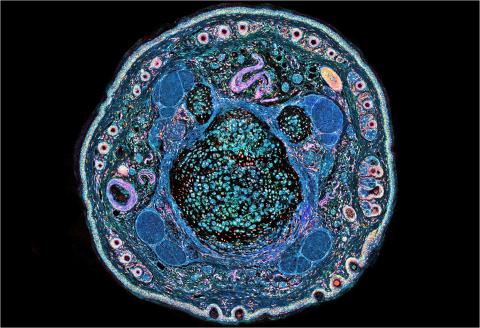
3395: NCMIR mouse tail
3395: NCMIR mouse tail
Stained cross section of a mouse tail.
Tom Deerinck, National Center for Microscopy and Imaging Research (NCMIR)
View Media

6764: Crystals of CCD-1 in complex with cefotaxime
6764: Crystals of CCD-1 in complex with cefotaxime
CCD-1 is an enzyme produced by the bacterium Clostridioides difficile that helps it resist antibiotics. Here, researchers crystallized bound pairs of CCD-1 molecules and molecules of the antibiotic cefotaxime. This enabled their structure to be studied using X-ray crystallography.
Related to images 6765, 6766, and 6767.
Related to images 6765, 6766, and 6767.
Keith Hodgson, Stanford University.
View Media
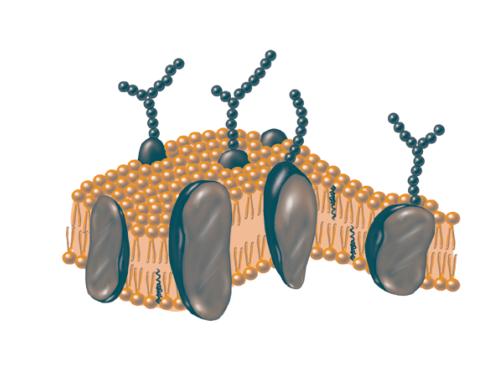
2523: Plasma membrane
2523: Plasma membrane
The plasma membrane is a cell's protective barrier. See image 2524 for a labeled version of this illustration. Featured in The Chemistry of Health.
Crabtree + Company
View Media
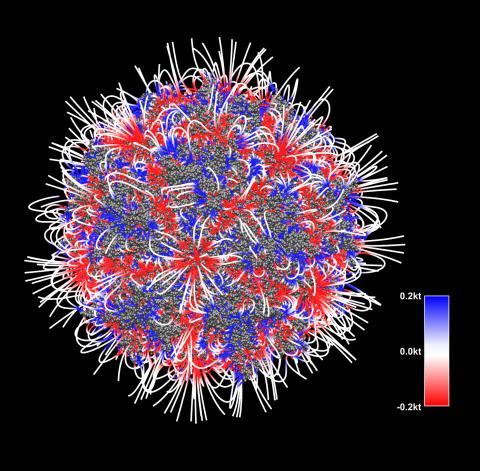
3375: Electrostatic map of the adeno-associated virus with scale
3375: Electrostatic map of the adeno-associated virus with scale
The new highly efficient parallelized DelPhi software was used to calculate the potential map distribution of an entire virus, the adeno-associated virus, which is made up of more than 484,000 atoms. Despite the relatively large dimension of this biological system, resulting in 815x815x815 mesh points, the parallelized DelPhi, utilizing 100 CPUs, completed the calculations within less than three minutes. Related to image 3374.
Emil Alexov, Clemson University
View Media
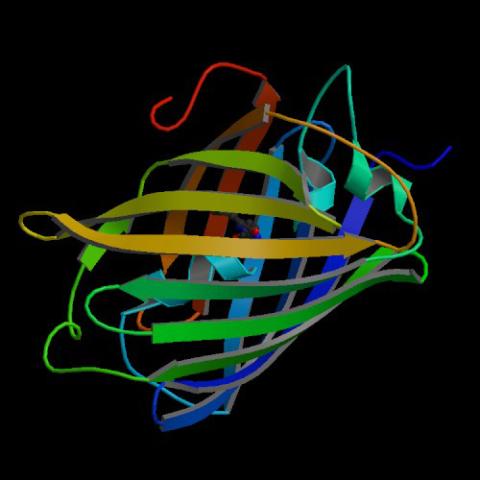
2317: Fruitful dyes
2317: Fruitful dyes
These colorful, computer-generated ribbons show the backbone of a molecule that glows a fluorescent red. The molecule, called mStrawberry, was created by chemists based on a protein found in the ruddy lips of a coral. Scientists use the synthetic molecule and other "fruity" ones like it as a dye to mark and study cell structures.
Roger Y. Tsien, University of California, San Diego
View Media
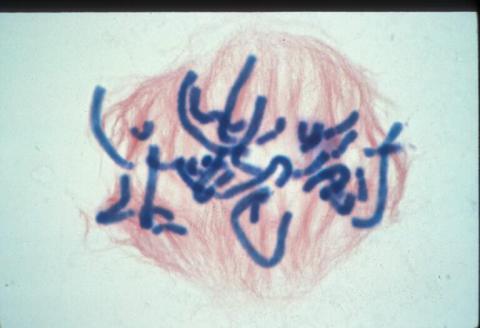
1016: Lily mitosis 06
1016: Lily mitosis 06
A light microscope image of a cell from the endosperm of an African globe lily (Scadoxus katherinae). This is one frame of a time-lapse sequence that shows cell division in action. The lily is considered a good organism for studying cell division because its chromosomes are much thicker and easier to see than human ones. Staining shows microtubules in red and chromosomes in blue. Here, condensed chromosomes are clearly visible and are starting to line up.
Related to images 1010, 1011, 1012, 1013, 1014, 1015, 1017, 1018, 1019, and 1021.
Related to images 1010, 1011, 1012, 1013, 1014, 1015, 1017, 1018, 1019, and 1021.
Andrew S. Bajer, University of Oregon, Eugene
View Media
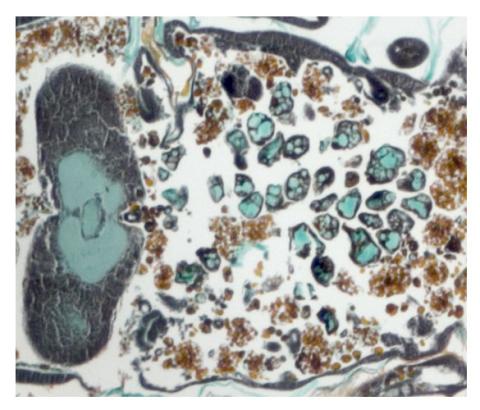
2759: Cross section of a Drosophila melanogaster pupa lacking Draper
2759: Cross section of a Drosophila melanogaster pupa lacking Draper
In the absence of the engulfment receptor Draper, salivary gland cells (light blue) persist in the thorax of a developing Drosophila melanogaster pupa. See image 2758 for a cross section of a normal pupa that does express Draper.
Christina McPhee and Eric Baehrecke, University of Massachusetts Medical School
View Media
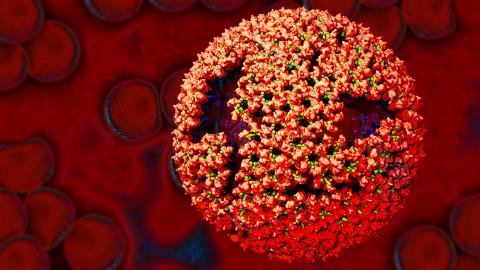
3771: Molecular model of freshly made Rous sarcoma virus (RSV)
3771: Molecular model of freshly made Rous sarcoma virus (RSV)
Viruses have been the foes of animals and other organisms for time immemorial. For almost as long, they've stayed well hidden from view because they are so tiny (they aren't even cells, so scientists call the individual virus a "particle"). This image shows a molecular model of a particle of the Rous sarcoma virus (RSV), a virus that infects and sometimes causes cancer in chickens. In the background is a photo of red blood cells. The particle shown is "immature" (not yet capable of infecting new cells) because it has just budded from an infected chicken cell and entered the bird's bloodstream. The outer shell of the immature virus is made up of a regular assembly of large proteins (shown in red) that are linked together with short protein molecules called peptides (green). This outer shell covers and protects the proteins (blue) that form the inner shell of the particle. But as you can see, the protective armor of the immature virus contains gaping holes. As the particle matures, the short peptides are removed and the large proteins rearrange, fusing together into a solid sphere capable of infecting new cells. While still immature, the particle is vulnerable to drugs that block its development. Knowing the structure of the immature particle may help scientists develop better medications against RSV and similar viruses in humans. Scientists used sophisticated computational tools to reconstruct the RSV atomic structure by crunching various data on the RSV proteins to simulate the entire structure of immature RSV.
Boon Chong Goh, University of Illinois at Urbana-Champaign
View Media
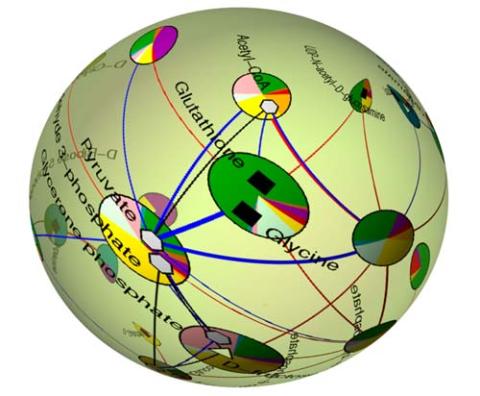
2331: Statistical cartography
2331: Statistical cartography
Like a world of its own, this sphere represents all the known chemical reactions in the E. coli bacterium. The colorful circles on the surface symbolize sets of densely interconnected reactions. The lines between the circles show additional connecting reactions. The shapes inside the circles are landmark molecules, like capital cities on a map, that either act as hubs for many groups of reactions, are highly conserved among species, or both. Molecules that connect far-flung reactions on the sphere are much more conserved during evolution than molecules that connect reactions within a single circle. This statistical cartography could reveal insights about other complex systems, such as protein interactions and gene regulation networks.
Luis A. Nunes Amaral, Northwestern University
View Media
2434: Fruit fly retina 02
2434: Fruit fly retina 02
Section of a fruit fly retina showing the light-sensing molecules rhodopsin-5 (blue) and rhodopsin-6 (red).
Hermann Steller, Rockefeller University
View Media
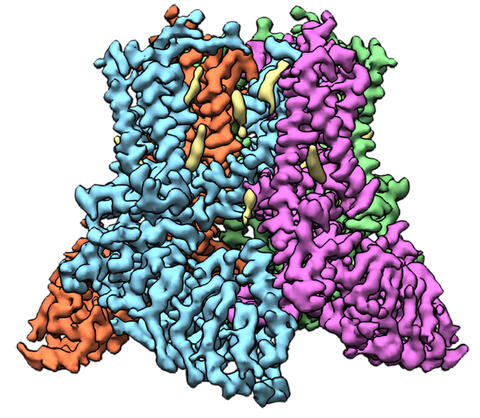
6577: Transient receptor potential channel TRPV5
6577: Transient receptor potential channel TRPV5
A 3D reconstruction of a transient receptor potential channel called TRPV5 that was created based on cryo-electron microscopy images. TRPV5 is primarily found in kidney cells and is essential for reabsorbing calcium into the blood.
Vera Moiseenkova-Bell, University of Pennsylvania.
View Media

6804: Staphylococcus aureus in the porous coating of a femoral hip stem
6804: Staphylococcus aureus in the porous coating of a femoral hip stem
Staphylococcus aureus bacteria (blue) on the porous coating of a femoral hip stem used in hip replacement surgery. The relatively rough surface of an implant is a favorable environment for bacteria to attach and grow. This can lead to the development of biofilms, which can cause infections. The researchers who took this image are working to understand where biofilms are likely to develop. This knowledge could support the prevention and treatment of infections. A scanning electron microscope was used to capture this image.
More information on the research that produced this image can be found in the Antibiotics paper "Free-floating aggregate and single-cell-initiated biofilms of Staphylococcus aureus" by Gupta et al.
Related to image 6803 and video 6805.
More information on the research that produced this image can be found in the Antibiotics paper "Free-floating aggregate and single-cell-initiated biofilms of Staphylococcus aureus" by Gupta et al.
Related to image 6803 and video 6805.
Paul Stoodley, The Ohio State University.
View Media
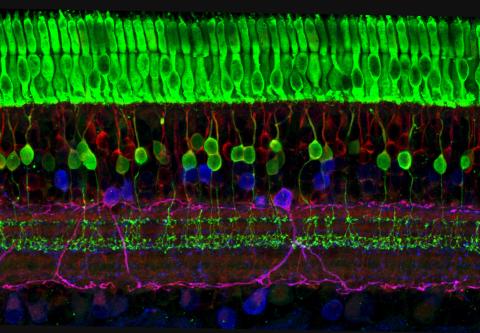
3635: The eye uses many layers of nerve cells to convert light into sight
3635: The eye uses many layers of nerve cells to convert light into sight
This image captures the many layers of nerve cells in the retina. The top layer (green) is made up of cells called photoreceptors that convert light into electrical signals to relay to the brain. The two best-known types of photoreceptor cells are rod- and cone-shaped. Rods help us see under low-light conditions but can't help us distinguish colors. Cones don't function well in the dark but allow us to see vibrant colors in daylight.
This image was part of the Life: Magnified exhibit that ran from June 3, 2014, to January 21, 2015, at Dulles International Airport.
This image was part of the Life: Magnified exhibit that ran from June 3, 2014, to January 21, 2015, at Dulles International Airport.
Wei Li, National Eye Institute, National Institutes of Health
View Media
2740: Early life of a protein
2740: Early life of a protein
This illustration represents the early life of a protein—specifically, apomyoglobin—as it is synthesized by a ribosome and emerges from the ribosomal tunnel, which contains the newly formed protein's conformation. The synthesis occurs in the complex swirl of the cell medium, filled with interactions among many molecules. Researchers in Silvia Cavagnero's laboratory are studying the structure and dynamics of newly made proteins and polypeptides using spectroscopic and biochemical techniques.
Silvia Cavagnero, University of Wisconsin, Madison
View Media
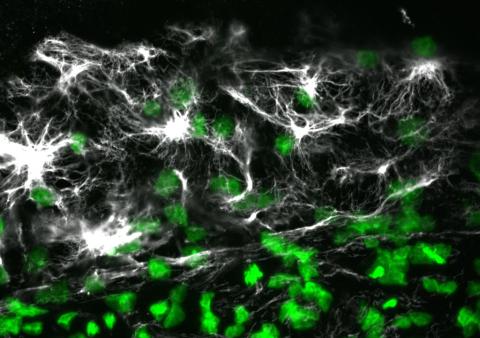
2807: Vimentin in a quail embryo
2807: Vimentin in a quail embryo
Confocal image showing high levels of the protein vimentin (white) at the edge zone of a quail embryo. Cell nuclei are labeled green. More specifically, this high-magnification (60X) image shows vimentin immunofluorescence in the edge zone (top of image) and inner zone (bottom of image) of a Stage 4 quail blastoderm. Vimentin expression (white) is shown merged with Sytox nuclear labeling (green) at the edge of the blastoderm. A thick vimentin filament runs circumferentially (parallel to the direction of the edge) that appears to delineate the transition between the edge zone and interior zone. Also shown are dense vimentin clusters or foci, which typically appear to be closely associated with edge cell nuclei. An NIGMS grant to Professor Garcia was used to purchase the confocal microscope that collected this image. Related to image 2808 and video 2809.
Andrés Garcia, Georgia Tech
View Media
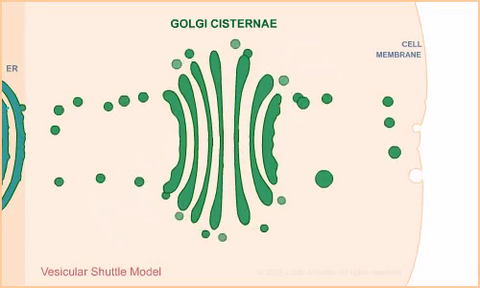
1306: Vesicular shuttle model
1306: Vesicular shuttle model
Animation for the vesicular shuttle model of Golgi transport.
Judith Stoffer
View Media
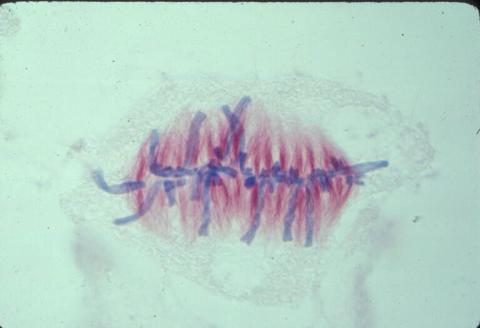
1017: Lily mitosis 07
1017: Lily mitosis 07
A light microscope image of a cell from the endosperm of an African globe lily (Scadoxus katherinae). This is one frame of a time-lapse sequence that shows cell division in action. The lily is considered a good organism for studying cell division because its chromosomes are much thicker and easier to see than human ones. Staining shows microtubules in red and chromosomes in blue. Here, condensed chromosomes are clearly visible and have lined up in the middle of the dividing cell.
Related to images 1010, 1011, 1012, 1013, 1014, 1015, 1016, 1018, 1019, and 1021.
Related to images 1010, 1011, 1012, 1013, 1014, 1015, 1016, 1018, 1019, and 1021.
Andrew S. Bajer, University of Oregon, Eugene
View Media
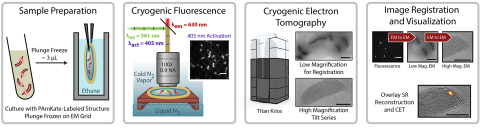
6568: Correlative imaging by annotation with single molecules (CIASM) process
6568: Correlative imaging by annotation with single molecules (CIASM) process
These images illustrate a technique combining cryo-electron tomography and super-resolution fluorescence microscopy called correlative imaging by annotation with single molecules (CIASM). CIASM enables researchers to identify small structures and individual molecules in cells that they couldn’t using older techniques.
Peter Dahlberg, Stanford University.
View Media
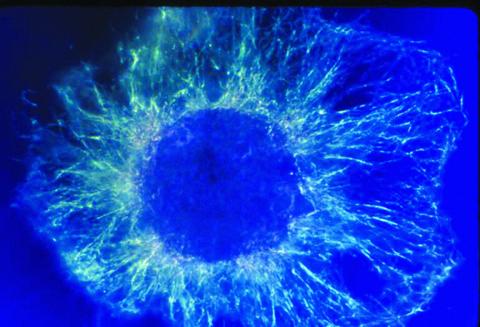
1058: Lily mitosis 01
1058: Lily mitosis 01
A light microscope image shows the chromosomes, stained dark blue, in a dividing cell of an African globe lily (Scadoxus katherinae). This is one frame of a time-lapse sequence that shows cell division in action. The lily is considered a good organism for studying cell division because its chromosomes are much thicker and easier to see than human ones.
Andrew S. Bajer, University of Oregon, Eugene
View Media
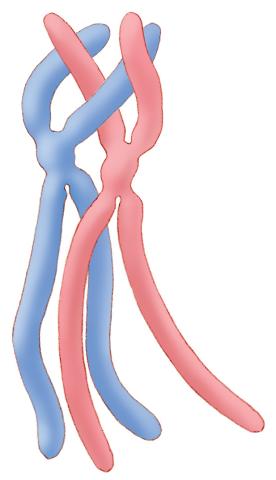
1315: Chromosomes before crossing over
1315: Chromosomes before crossing over
Duplicated pair of chromosomes lined up and ready to cross over.
Judith Stoffer
View Media
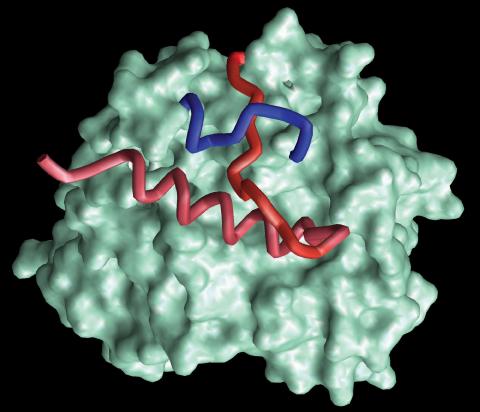
2374: Protein from Methanobacterium thermoautotrophicam
2374: Protein from Methanobacterium thermoautotrophicam
A knotted protein from an archaebacterium called Methanobacterium thermoautotrophicam. This organism breaks down waste products and produces methane gas. Protein folding theory previously held that forming a knot was beyond the ability of a protein, but this structure, determined at Argonne's Structural Biology Center, proves differently. Researchers theorize that this knot stabilizes the amino acid subunits of the protein.
Midwest Center For Structural Genomics, PSI
View Media
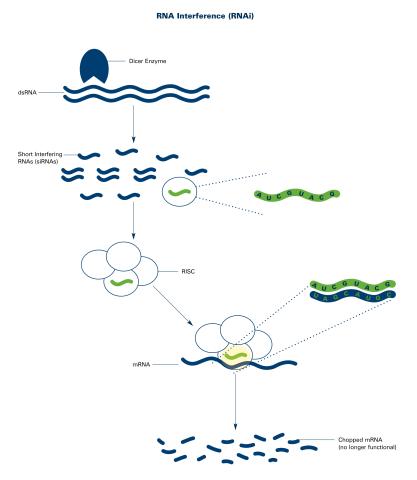
2559: RNA interference (with labels)
2559: RNA interference (with labels)
RNA interference or RNAi is a gene-silencing process in which double-stranded RNAs trigger the destruction of specific RNAs. See 2558 for an unlabeled version of this illustration. Featured in The New Genetics.
Crabtree + Company
View Media
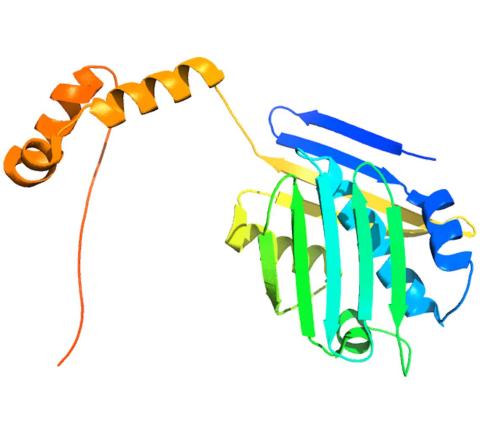
3355: Hsp33 figure 2
3355: Hsp33 figure 2
Featured in the March 15, 2012 issue of Biomedical Beat. Related to Hsp33 Figure 1, image 3354.
Ursula Jakob and Dana Reichmann, University of Michigan
View Media
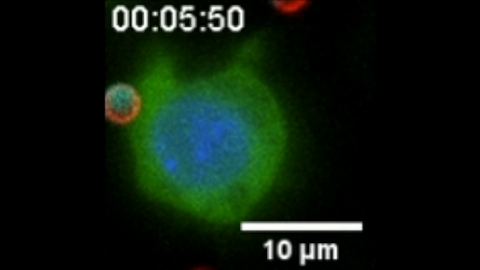
6801: “Two-faced” Janus particle activating a macrophage
6801: “Two-faced” Janus particle activating a macrophage
A macrophage—a type of immune cell that engulfs invaders—“eats” and is activated by a “two-faced” Janus particle. The particle is called “two-faced” because each of its two hemispheres is coated with a different type of molecule, shown here in red and cyan. During macrophage activation, a transcription factor tagged with a green fluorescence protein (NF-κB) gradually moves from the cell’s cytoplasm into its nucleus and causes DNA transcription. The distribution of molecules on “two-faced” Janus particles can be altered to control the activation of immune cells. Details on this “geometric manipulation” strategy can be found in the Proceedings of the National Academy of Sciences paper "Geometrical reorganization of Dectin-1 and TLR2 on single phagosomes alters their synergistic immune signaling" by Li et al. and the Scientific Reports paper "Spatial organization of FcγR and TLR2/1 on phagosome membranes differentially regulates their synergistic and inhibitory receptor crosstalk" by Li et al. This video was captured using epi-fluorescence microscopy.
Related to video 6800.
Related to video 6800.
Yan Yu, Indiana University, Bloomington.
View Media
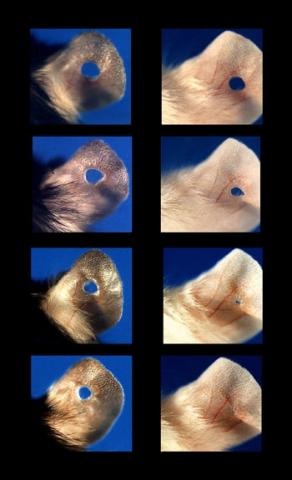
3426: Regeneration of Mouse Ears
3426: Regeneration of Mouse Ears
Normal mice, like the B6 breed pictured on the left, develop scars when their ears are pierced. The Murphy Roths Large (MRL) mice pictured on the right can grow back lost ear tissue thanks to an inactive version of the p21 gene. When researchers knocked out that same gene in other mouse breeds, their ears also healed completely without scarring. Journal Article: Clark, L.D., Clark, R.K. and Heber-Katz, E. 1998. A new murine model for mammalian wound repair and regeneration. Clin Immunol Immunopathol 88: 35-45.
Ellen Heber-Katz, The Wistar Institute
View Media

2396: Hen egg lysozyme (1)
2396: Hen egg lysozyme (1)
Crystals of hen egg lysozyme protein created for X-ray crystallography, which can reveal detailed, three-dimensional protein structures.
Alex McPherson, University of California, Irvine
View Media
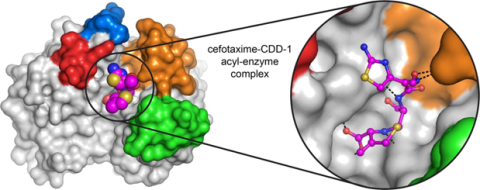
6767: Space-filling model of a cefotaxime-CCD-1 complex
6767: Space-filling model of a cefotaxime-CCD-1 complex
CCD-1 is an enzyme produced by the bacterium Clostridioides difficile that helps it resist antibiotics. Using X-ray crystallography, researchers determined the structure of a complex between CCD-1 and the antibiotic cefotaxime (purple, yellow, and blue molecule). The structure revealed that CCD-1 provides extensive hydrogen bonding (shown as dotted lines) and stabilization of the antibiotic in the active site, leading to efficient degradation of the antibiotic.
Related to images 6764, 6765, and 6766.
Related to images 6764, 6765, and 6766.
Keith Hodgson, Stanford University.
View Media

2385: Heat shock protein complex from Methanococcus jannaschii
2385: Heat shock protein complex from Methanococcus jannaschii
Model based on X-ray crystallography of the structure of a small heat shock protein complex from the bacteria, Methanococcus jannaschii. Methanococcus jannaschii is an organism that lives at near boiling temperature, and this protein complex helps it cope with the stress of high temperature. Similar complexes are produced in human cells when they are "stressed" by events such as burns, heart attacks, or strokes. The complexes help cells recover from the stressful event.
Berkeley Structural Genomics Center, PSI-1
View Media
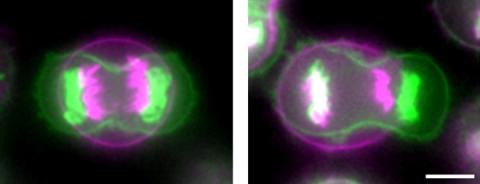
3648: Symmetrically and asymmetrically elongating cells
3648: Symmetrically and asymmetrically elongating cells
Merged fluorescent images of symmetrically (left) or asymmetrically (right) elongating HeLa cells at the end of early anaphase (magenta) and late anaphase (green). Chromosomes and cortical actin are visualized by expressing mCherry-histone H2B and Lifeact-mCherry. Scale bar, 10µm. See the PubMed abstract of this research.
Tomomi Kiyomitsu and Iain M. Cheeseman, Whitehead Institute for Biomedical Research
View Media
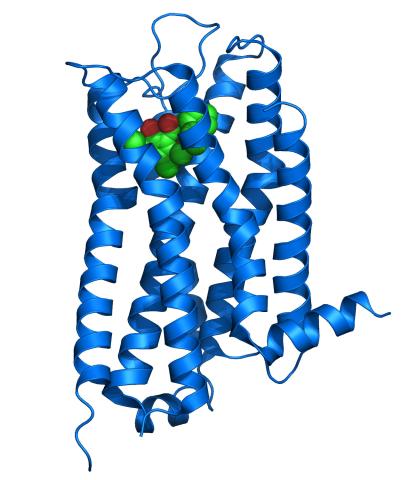
3363: Dopamine D3 receptor
3363: Dopamine D3 receptor
The receptor is shown bound to an antagonist, eticlopride
Raymond Stevens, The Scripps Research Institute
View Media

2509: From DNA to Protein
2509: From DNA to Protein
Nucleotides in DNA are copied into RNA, where they are read three at a time to encode the amino acids in a protein. Many parts of a protein fold as the amino acids are strung together.
See image 2510 for a labeled version of this illustration.
Featured in The Structures of Life.
See image 2510 for a labeled version of this illustration.
Featured in The Structures of Life.
Crabtree + Company
View Media
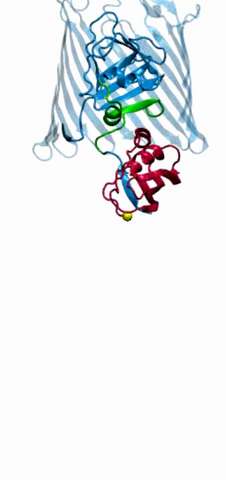
2304: Bacteria working to eat
2304: Bacteria working to eat
Gram-negative bacteria perform molecular acrobatics just to eat. Because they're encased by two membranes, they must haul nutrients across both. To test one theory of how the bacteria manage this feat, researchers used computer simulations of two proteins involved in importing vitamin B12. Here, the protein (red) anchored in the inner membrane of bacteria tugs on a much larger protein (green and blue) in the outer membrane. Part of the larger protein unwinds, creating a pore through which the vitamin can pass.
Emad Tajkhorshid, University of Illinois at Urbana-Champaign
View Media
5811: NCMIR Tongue 2
5811: NCMIR Tongue 2
Microscopy image of a tongue. One in a series of two, see image 5810
National Center for Microscopy and Imaging Research (NCMIR)
View Media
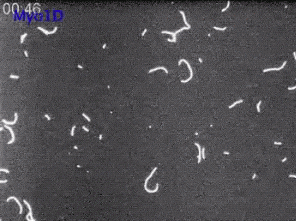
6562: Drosophila (fruit fly) myosin 1D motility assay
6562: Drosophila (fruit fly) myosin 1D motility assay
Actin gliding powered by myosin 1D. Note the counterclockwise motion of the gliding actin filaments.
Serapion Pyrpassopoulos and E. Michael Ostap, University of Pennsylvania
View Media
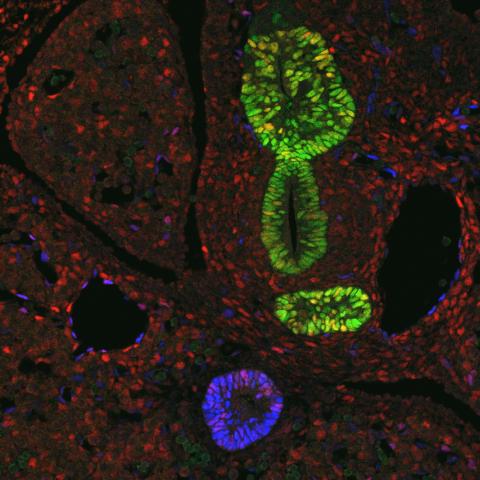
3440: Transcription factor Sox17 controls embryonic development of certain internal organs
3440: Transcription factor Sox17 controls embryonic development of certain internal organs
During embryonic development, transcription factors (proteins that regulate gene expression) govern the differentiation of cells into separate tissues and organs. Researchers at Cincinnati Children's Hospital Medical Center used mice to study the development of certain internal organs, including the liver, pancreas, duodenum (beginning part of the small intestine), gall bladder and bile ducts. They discovered that transcription factor Sox17 guides some cells to develop into liver cells and others to become part of the pancreas or biliary system (gall bladder, bile ducts and associated structures). The separation of these two distinct cell types (liver versus pancreas/biliary system) is complete by embryonic day 8.5 in mice. The transcription factors PDX1 and Hes1 are also known to be involved in embryonic development of the pancreas and biliary system. This image shows mouse cells at embryonic day 10.5. The green areas show cells that will develop into the pancreas and/or duodenum(PDX1 is labeled green). The blue area near the bottom will become the gall bladder and the connecting tubes (common duct and cystic duct) that attach the gall bladder to the liver and pancreas (Sox17 is labeled blue). The transcription factor Hes1 is labeled red. The image was not published. A similar image (different plane of the section) was published in: Sox17 Regulates Organ Lineage Segregation of Ventral Foregut Progenitor Cells Jason R. Spence, Alex W. Lange, Suh-Chin J. Lin, Klaus H. Kaestner, Andrew M. Lowy, Injune Kim, Jeffrey A. Whitsett and James M. Wells, Developmental Cell, Volume 17, Issue 1, 62-74, 21 July 2009. doi:10.1016/j.devcel.2009.05.012
James M. Wells, Cincinnati Children's Hospital Medical Center
View Media
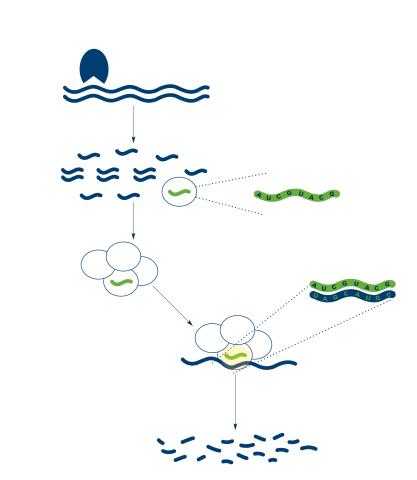
2558: RNA interference
2558: RNA interference
RNA interference or RNAi is a gene-silencing process in which double-stranded RNAs trigger the destruction of specific RNAs. See 2559 for a labeled version of this illustration. Featured in The New Genetics.
Crabtree + Company
View Media
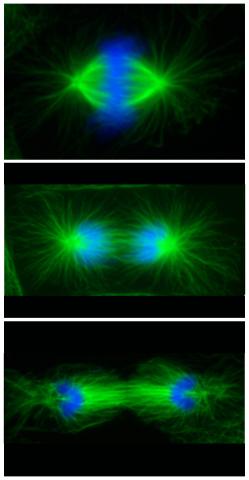
3442: Cell division phases in Xenopus frog cells
3442: Cell division phases in Xenopus frog cells
These images show three stages of cell division in Xenopus XL177 cells, which are derived from tadpole epithelial cells. They are (from top): metaphase, anaphase and telophase. The microtubules are green and the chromosomes are blue. Related to 3443.
Claire Walczak, who took them while working as a postdoc in the laboratory of Timothy Mitchison
View Media
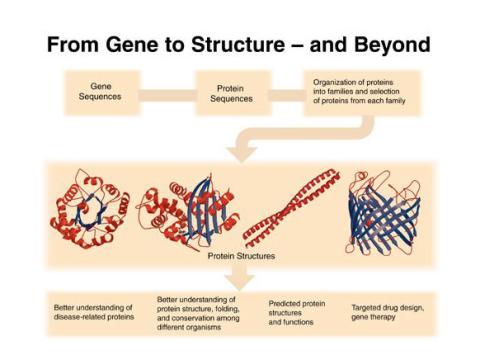
2363: PSI: from genes to structures
2363: PSI: from genes to structures
The goal of the Protein Structure Initiative (PSI) is to determine the three-dimensional shapes of a wide range of proteins by solving the structures of representative members of each protein family found in nature. The collection of structures should serve as a valuable resource for biomedical research scientists.
National Institute of General Medical Sciences
View Media
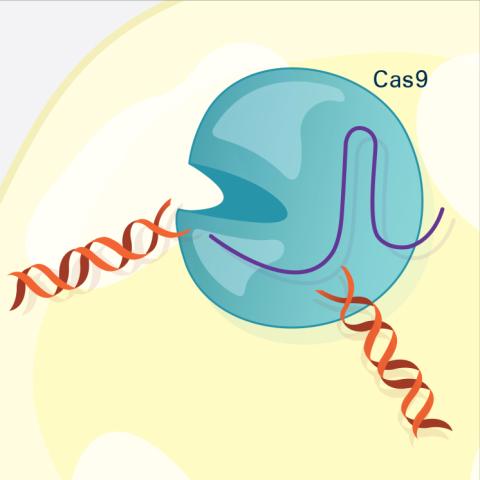
6487: CRISPR Illustration Frame 3
6487: CRISPR Illustration Frame 3
This illustration shows, in simplified terms, how the CRISPR-Cas9 system can be used as a gene-editing tool. The CRISPR system has two components joined together: a finely tuned targeting device (a small strand of RNA programmed to look for a specific DNA sequence) and a strong cutting device (an enzyme called Cas9 that can cut through a double strand of DNA). In this frame (3 of 4), the Cas9 enzyme cuts both strands of the DNA.
For an explanation and overview of the CRISPR-Cas9 system, see the iBiology video, and find the full CRIPSR illustration here.
For an explanation and overview of the CRISPR-Cas9 system, see the iBiology video, and find the full CRIPSR illustration here.
National Institute of General Medical Sciences.
View Media

1069: Lab mice
1069: Lab mice
Many researchers use the mouse (Mus musculus) as a model organism to study mammalian biology. Mice carry out practically all the same life processes as humans and, because of their small size and short generation times, are easily raised in labs. Scientists studying a certain cellular activity or disease can choose from tens of thousands of specially bred strains of mice to select those prone to developing certain tumors, neurological diseases, metabolic disorders, premature aging, or other conditions.
Bill Branson, National Institutes of Health
View Media

3724: Snowflake DNA origami
3724: Snowflake DNA origami
An atomic force microscopy image shows DNA folded into an intricate, computer-designed structure. The image is featured on Biomedical Beat blog post Cool Images: A Holiday-Themed Collection. For more background on DNA origami, see Cool Image: DNA Origami. See also related image 3690.
Hao Yan, Arizona State University
View Media
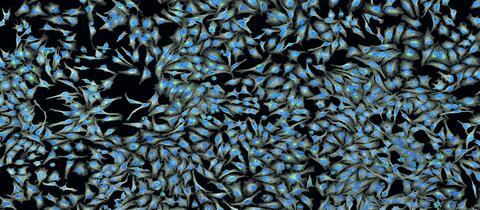
5761: A panorama view of cells
5761: A panorama view of cells
This photograph shows a panoramic view of HeLa cells, a cell line many researchers use to study a large variety of important research questions. The cells' nuclei containing the DNA are stained in blue and the cells' cytoskeletons in gray.
Tom Deerinck, National Center for Microscopy and Imaging Research
View Media
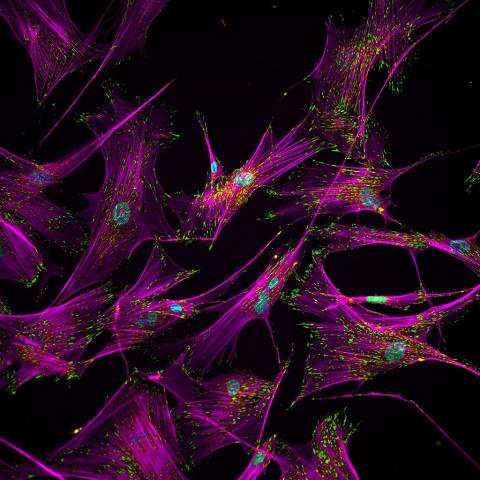
3457: Sticky stem cells
3457: Sticky stem cells
Like a group of barnacles hanging onto a rock, these human cells hang onto a matrix coated glass slide. Actin stress fibers, stained magenta, and the protein vinculin, stained green, make this adhesion possible. The fibroblast nuclei are stained blue.
Ankur Singh and Andrés García, Georgia Institute of Technology
View Media